The Nytricx Nitric Oxide (NO)-Releasing Wound Patch: Controlled Antimicrobial and Wound Healing Therapy

Nytricx Wound Patch Platform: Product Variants
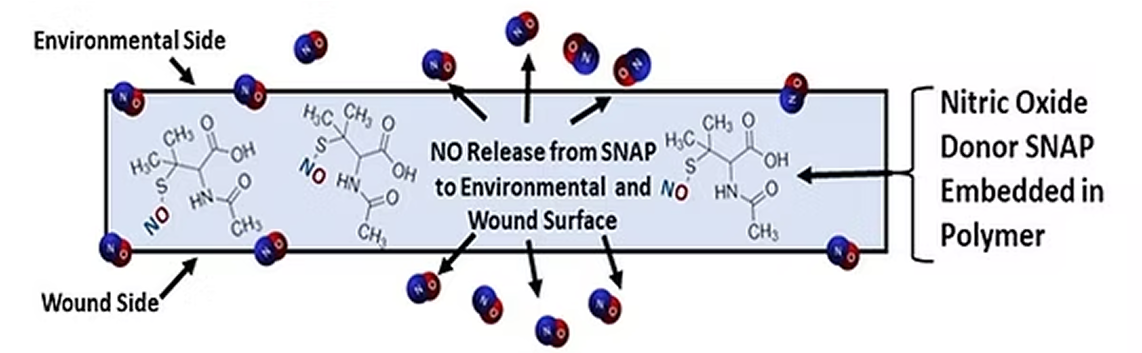
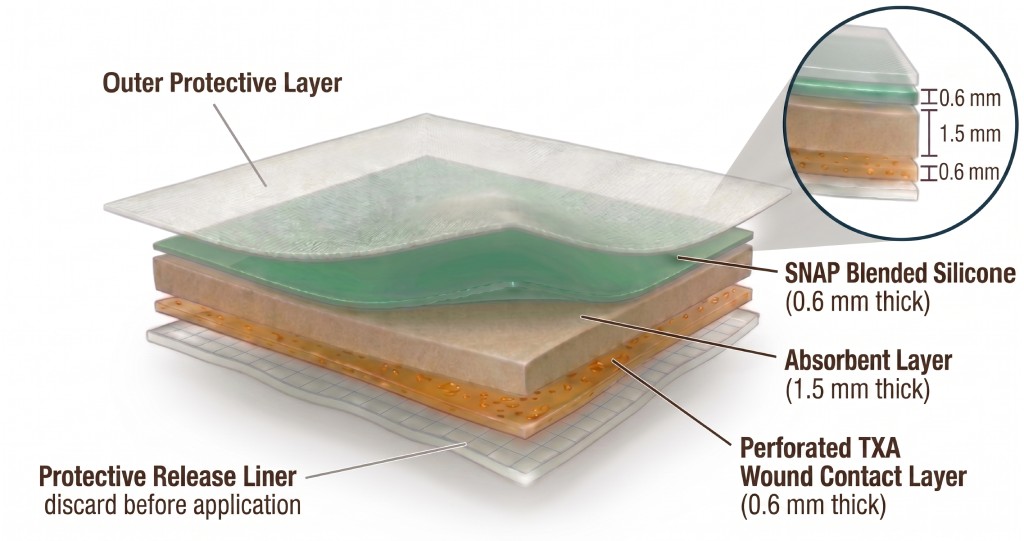
Nitric Oxide-releasing wound patch
Ideal for: Chronic wounds, diabetic foot ulcers, surgical sites
The core dressing technology uses medical-grade silicone embedded with SNAP (a nitric oxide donor). It continuously releases NO to both the wound surface and environment, helping prevent infection, reduce inflammation, and promote tissue regeneration.
- Continuous NO release
- Thin, flexible, and non-cytotoxic
- No demonstrated antibiotic resistance risk
Nitric Oxide-releasing wound patch
Ideal for: Chronic wounds, diabetic foot ulcers, surgical sites
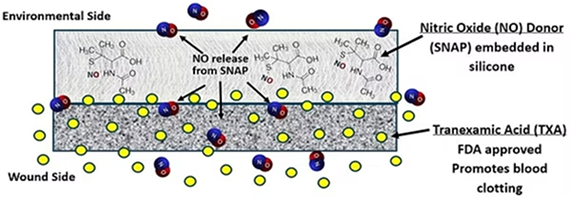
Dual Nitric Oxide-releasing and blood clotting wound patch
Ideal for: Emergency medicine, trauma wounds, battlefield injury care
Dual Nitric Oxide-releasing and blood clotting wound patch
Ideal for: Emergency medicine, trauma wounds, battlefield injury care
This dual-layer patch adds a second layer containing Tranexamic Acid (TXA), an FDA-approved clotting agent, while maintaining antimicrobial NO release. Designed for trauma, battlefield use, and post-surgical bleeding risk.
- Combines antimicrobial and hemostatic action
- TXA layer for rapid clotting
- Maintains NO release effectiveness
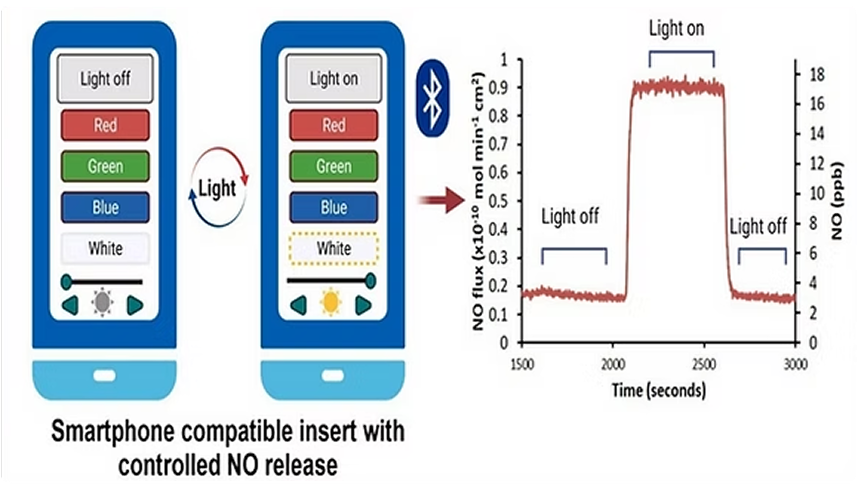
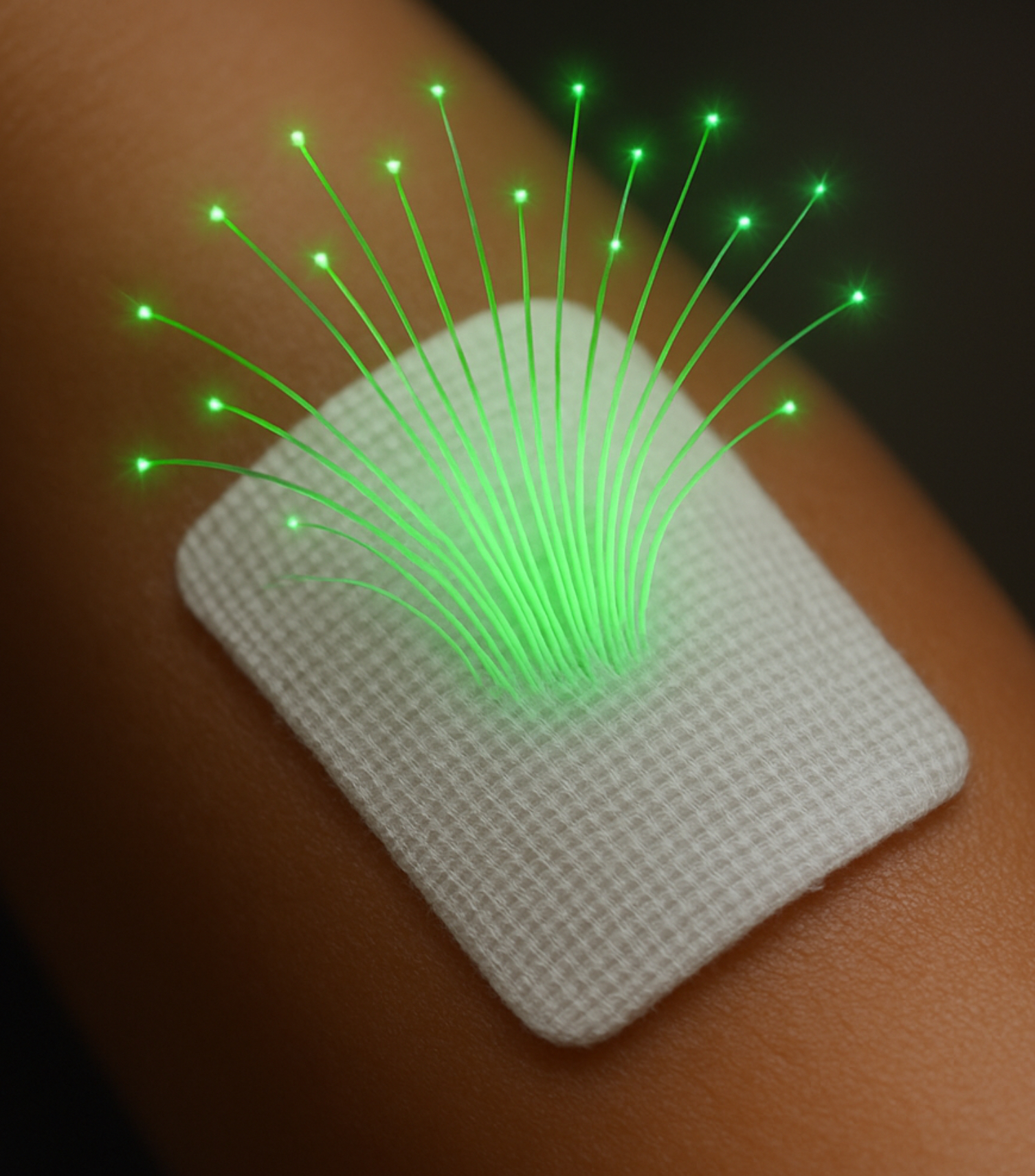
Fiber Optic Controlled Nitric Oxide-Releasing Wound Patch
Ideal for: Chronic Wound Therapy
This advanced version integrates a fiber optic element that enables real-time control of NO release through light modulation. NO output is tunable based on light intensity, offering precision controlled release of NO.
- Real-time NO control via light intensity
- Compatible with wearable tech/smartphone activation
- Ideal for long-term or complex wound management
Fiber Optic Controlled Nitric Oxide-Releasing Wound Patch
Ideal for: Controlled wound therapy, hospital-managed chronic wound care, R&D use cases

Scientific Highlights
-
Broad Antimicrobial Action
99.9% MRSA reduction in 3 days (porcine wound model)
-
Promotes All Healing Phases
Supports hemostasis, inflammation damping, proliferation, and remodeling
-
Predictable, Tunable Release
Fiber-optic version allows light-controlled NO dosing for precision therapy
Pre‑Clinical Data Snapshot
- Model
Gold-standard porcine dermal wounds
- Results:
3-log MRSA reduction in 3 days Enhanced re-epithelialization & moist healing
- Mechanism
NO: broad-spectrum antimicrobial + anti-inflammatory + pro-angiogenic
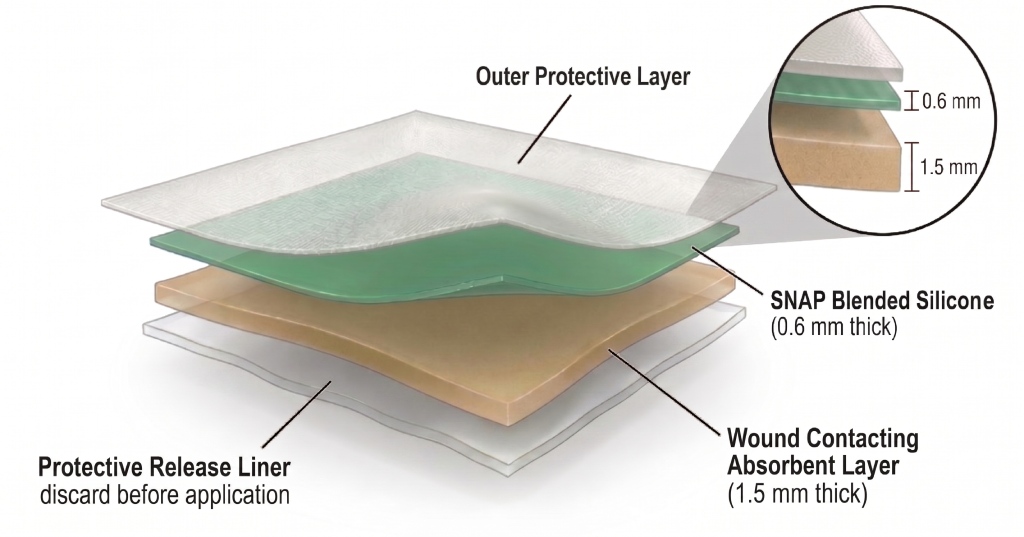
Design & Composition
- Medical-grade silicone (NuSil®) base
- Embedded SNAP for sustained NO release
- (Optional) Dual-layer version includes Tranexamic Acid for clotting
- NO-releasing Fiber Optic Wound Patch
Four stages of Wound Healing Regulated/
Promoted by Nitric Oxide
Hemostasis
NO Regulates
- Thrombosis
- Proliferation
- Adhesion expression
Inflammation
NO Regulates
- Leukocyte infiltration
Proliferation
NO Promotes
- Fibroblast & endothelial cell proliferation
- Collagen synthesis
- Angiogenesis
Remodeling
NO Promotes
- Increased collagen synthesis
- Collagen reorganization & reorientataion
Commercial Outlook & Strategic Development
Market Opportunity
The global wound care market is projected to exceed $30 billion by 2028, driven by rising cases of chronic wounds, surgical site infections, and diabetic foot ulcers.
- 19 million+ diabetic foot ulcer (DFU) cases worldwide
- Significant demand for non-antibiotic, antimicrobial wound technologies
Funding Secured
Nytricx has raised $6.2 million to date in non-dilutive government grants from – CDC, NIH, DoD, and BARDA
An additional $6.3 million in NIH funding is currently pending.
Seeking Strategic Partners
We are currently raising $10 million in milestone-based Series A funding to advance our NO-releasing wound patch to clinical deployment.
| Milestone | Use of Funds | Amount | Target Year |
|---|---|---|---|
| 1. | Finalize formulation & non-GLP safety evaluation | $2 Million | 2026 |
| 2. | GMP manufacturing & GLP toxicology studies | $4 Million | 2027 |
| 3. | Phase II clinical trials | $4 Million | 2028 |
Flexible terms available. Interested in investing or partnering?
Contact us to schedule a conversation.
Contact Us
President and CEO